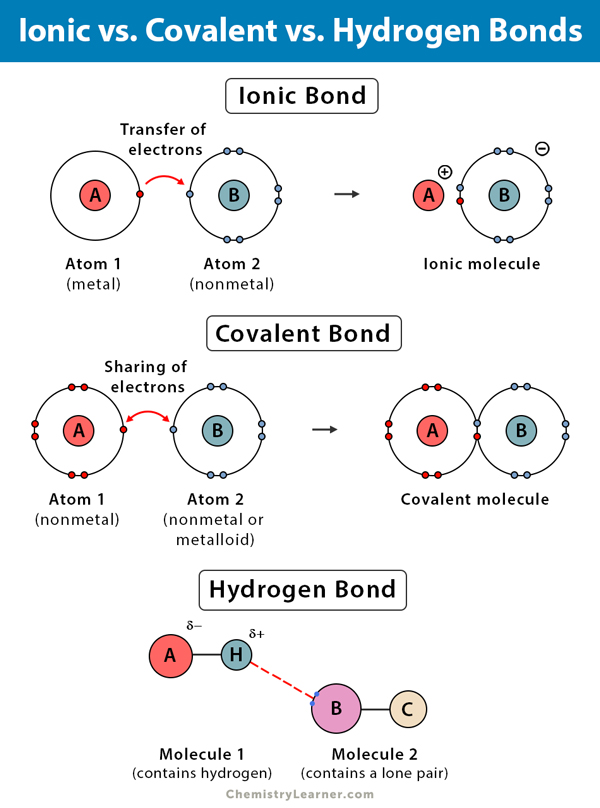
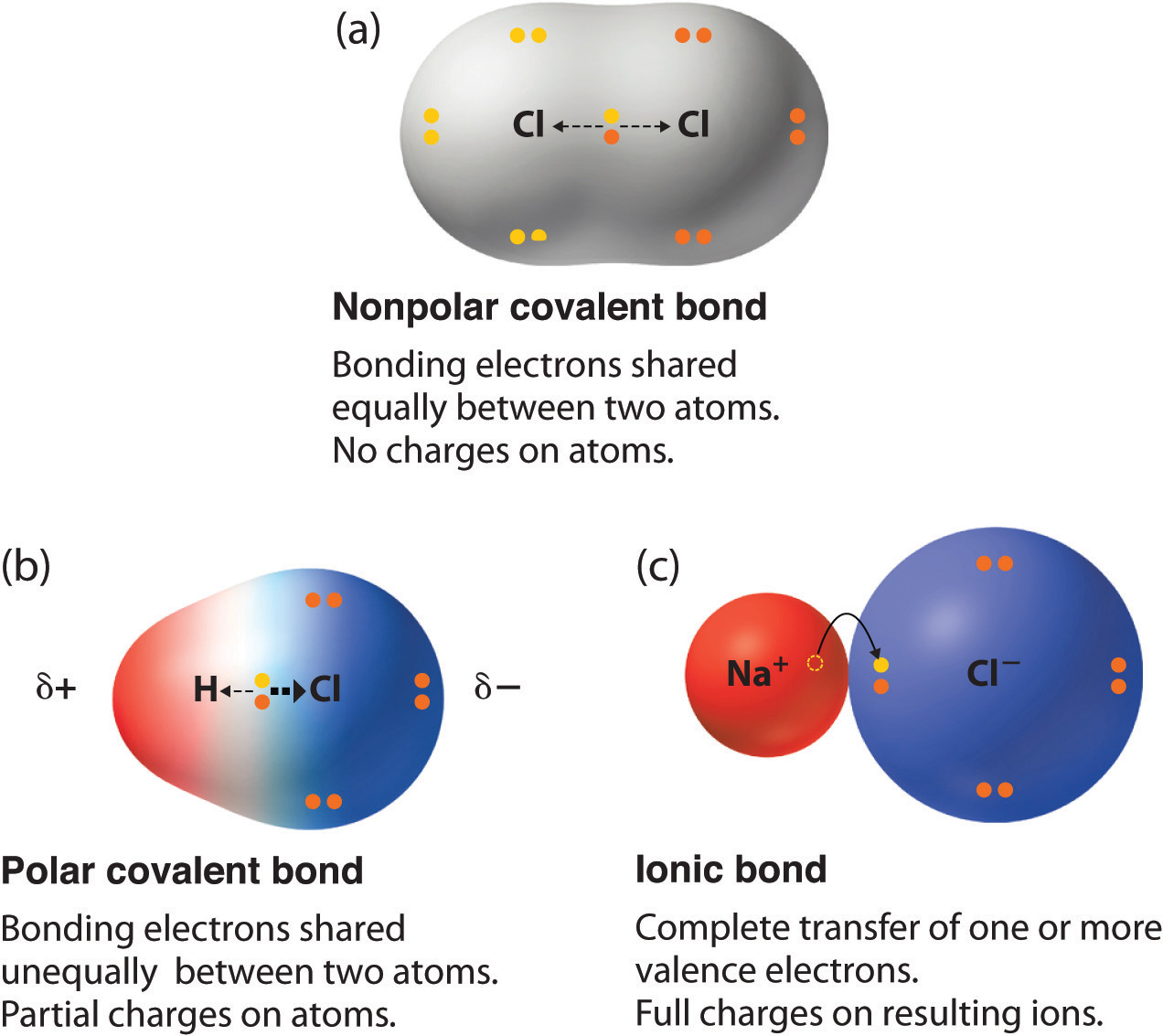
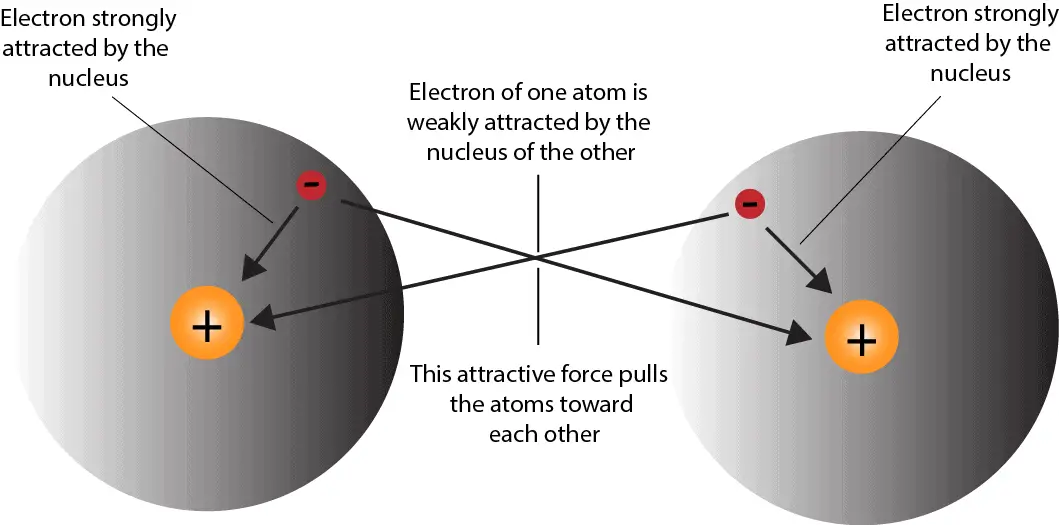
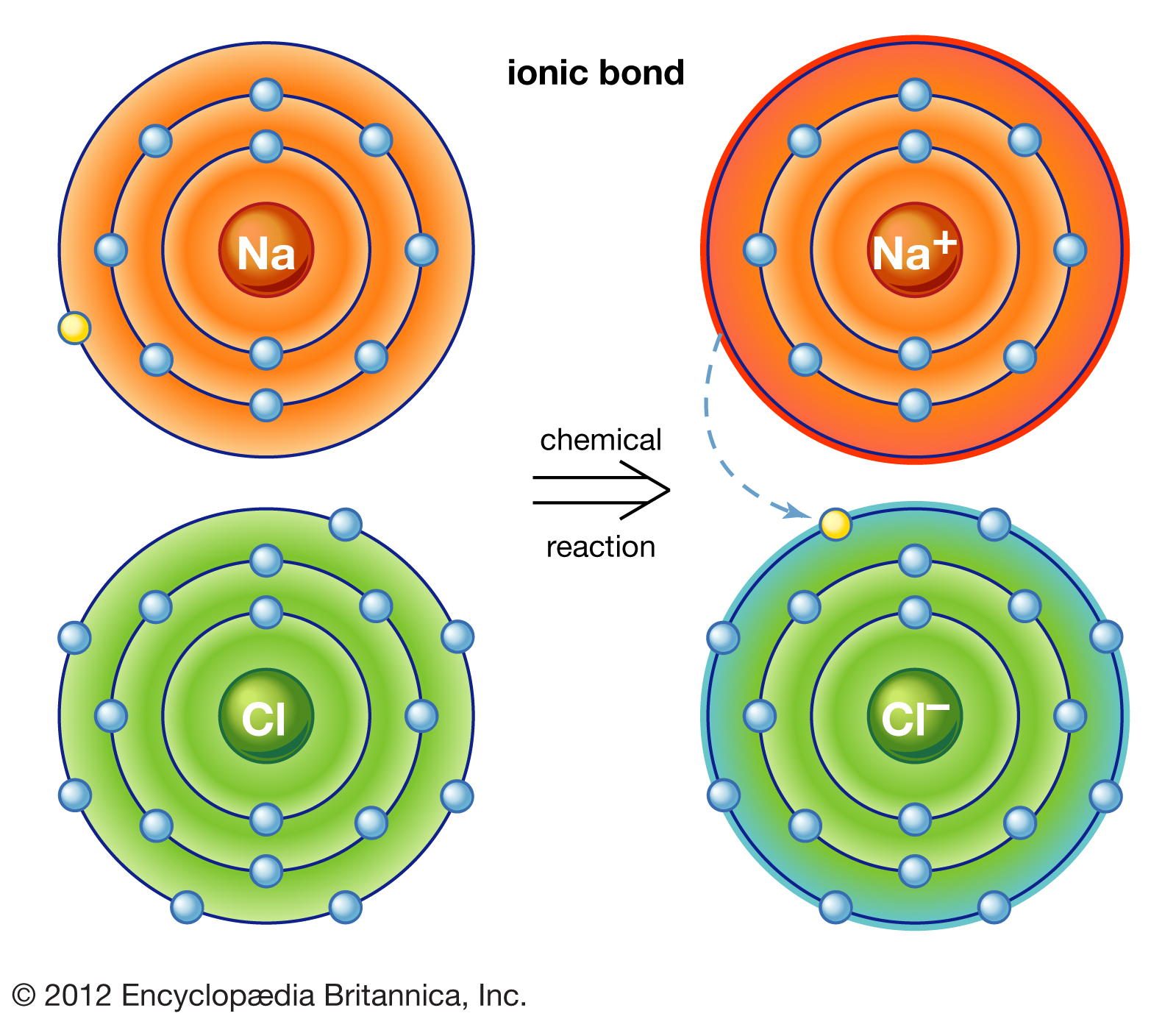
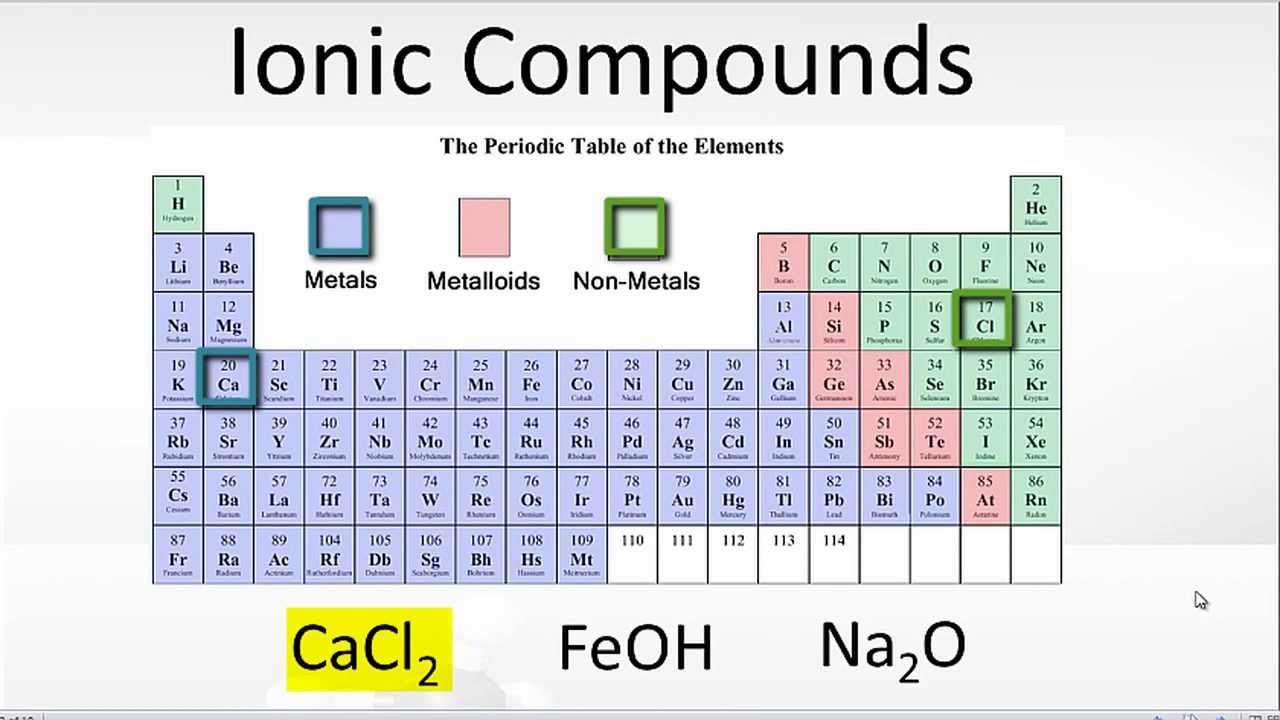
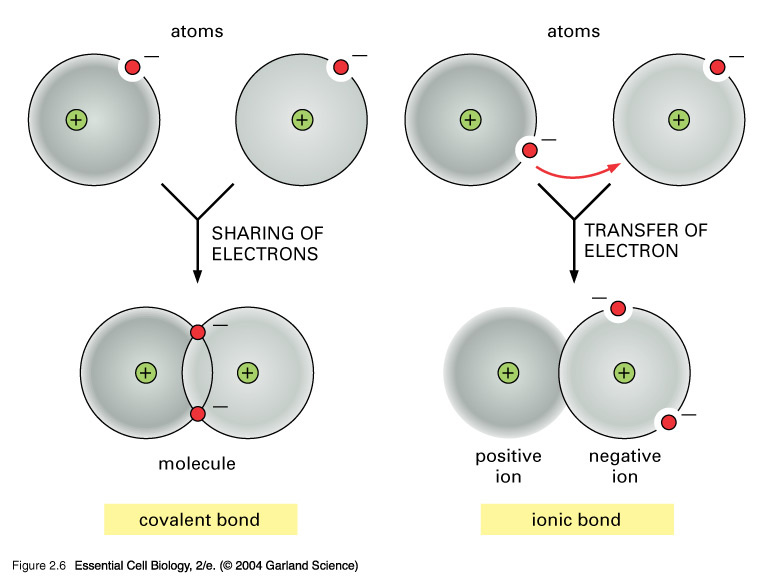
Why Do Atoms Form Ionic And Covalent Bonds - Ionic bonds require at least one electron. Because opposite charges attract (while like charges repel), these. Charged atoms are called ions. In ionic bonding, atoms transfer electrons to each other.
In ionic bonding, atoms transfer electrons to each other. Charged atoms are called ions. Because opposite charges attract (while like charges repel), these. Ionic bonds require at least one electron.
Ionic bonds require at least one electron. Charged atoms are called ions. In ionic bonding, atoms transfer electrons to each other. Because opposite charges attract (while like charges repel), these.
Difference Between Covalent and Ionic Bonds
In ionic bonding, atoms transfer electrons to each other. Charged atoms are called ions. Ionic bonds require at least one electron. Because opposite charges attract (while like charges repel), these.
Covalent Bond Is Between Covalent Bonds vs. Ionic Bonds What is The
Ionic bonds require at least one electron. In ionic bonding, atoms transfer electrons to each other. Charged atoms are called ions. Because opposite charges attract (while like charges repel), these.
Chapter 5.6 Properties of Polar Covalent Bonds Chemistry LibreTexts
Charged atoms are called ions. In ionic bonding, atoms transfer electrons to each other. Because opposite charges attract (while like charges repel), these. Ionic bonds require at least one electron.
Compounds With Both Ionic and Covalent Bonds
Ionic bonds require at least one electron. Charged atoms are called ions. In ionic bonding, atoms transfer electrons to each other. Because opposite charges attract (while like charges repel), these.
How do atoms form covalent bond?
Charged atoms are called ions. In ionic bonding, atoms transfer electrons to each other. Ionic bonds require at least one electron. Because opposite charges attract (while like charges repel), these.
Reading Covalent Bonds Biology I
Because opposite charges attract (while like charges repel), these. In ionic bonding, atoms transfer electrons to each other. Ionic bonds require at least one electron. Charged atoms are called ions.
chemistry knowledge Comparison between Covalent and Ionic Bond
Ionic bonds require at least one electron. Because opposite charges attract (while like charges repel), these. In ionic bonding, atoms transfer electrons to each other. Charged atoms are called ions.
How To Form Ionic Bonds
Charged atoms are called ions. In ionic bonding, atoms transfer electrons to each other. Ionic bonds require at least one electron. Because opposite charges attract (while like charges repel), these.
Covalent bond Definition, Properties, Examples, & Facts Britannica
Charged atoms are called ions. Because opposite charges attract (while like charges repel), these. Ionic bonds require at least one electron. In ionic bonding, atoms transfer electrons to each other.
In Ionic Bonding, Atoms Transfer Electrons To Each Other.
Because opposite charges attract (while like charges repel), these. Ionic bonds require at least one electron. Charged atoms are called ions.